|
Conclusionĭalton's Atomic Theory was a significant milestone in the development of atomic theory, providing a basic framework for understanding the behavior of matter. 
The conservation of atoms is now understood in terms of the conservation of mass and the conservation of charge. However, the theory expands upon this concept by explaining that chemical reactions involve the rearrangement of electrons, leading to the formation of new compounds. This principle still holds true in Modern Atomic Theory. Conservation of Atomsĭalton's theory stated that atoms are neither created nor destroyed in chemical reactions. The probabilistic nature of electron behavior is a fundamental aspect of quantum mechanics. These orbitals define the energy levels and sublevels where electrons are likely to be found. 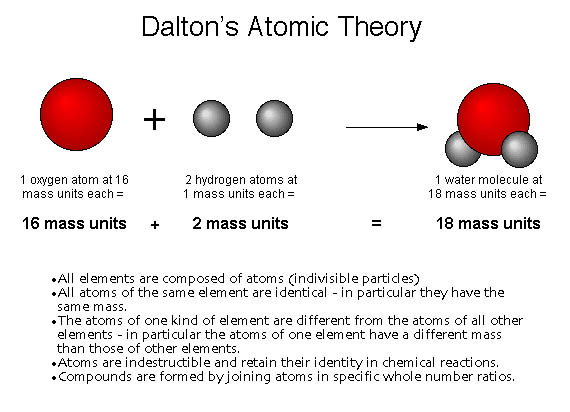
Modern Atomic Theory, on the other hand, recognizes that electrons do not follow precise paths around the nucleus but exist in regions of high probability called orbitals. Electron Behaviorĭalton's theory did not address the behavior of electrons in detail. This model explains the stability of atoms and the behavior of electrons in chemical reactions. In contrast, Modern Atomic Theory describes the presence of a nucleus containing protons and neutrons, surrounded by electrons occupying specific energy levels or orbitals. Atomic Structureĭalton's theory did not provide any insights into the internal structure of atoms. Isotopes have the same atomic number but different mass numbers, leading to variations in their physical and chemical properties. However, Modern Atomic Theory has revealed that atoms of the same element can have different numbers of neutrons, resulting in isotopes. Atomic Identityĭalton's theory stated that atoms of the same element are identical in size, mass, and other properties. The discovery of these particles has provided a deeper understanding of atomic structure and behavior. While this concept still holds true in Modern Atomic Theory, we now know that atoms are further composed of subatomic particles, including protons, neutrons, and electrons. Composition of Matterĭalton's theory proposed that all matter is composed of indivisible atoms. 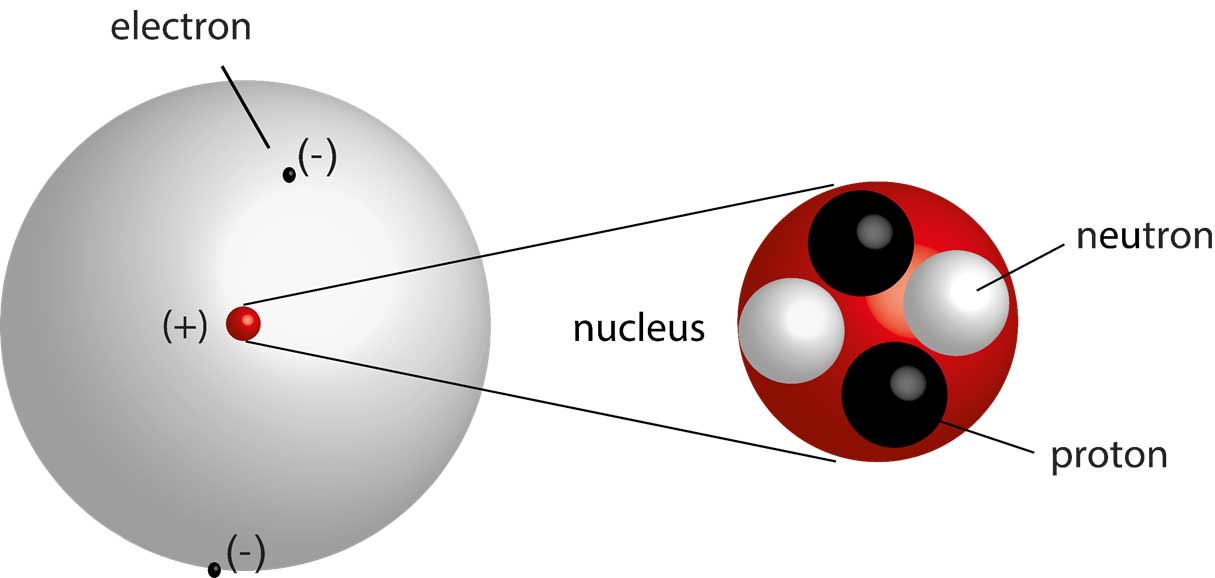
Let's compare the attributes of both theories in more detail: 1. While Dalton's Atomic Theory laid the foundation for our understanding of atoms, Modern Atomic Theory has expanded upon and refined many of its postulates. This theory has been extensively supported by experimental evidence and mathematical models. It takes into account the wave-particle duality of electrons and the probabilistic nature of their positions within an atom. Modern Atomic Theory provides a more detailed and accurate understanding of atomic structure and behavior. Chemical reactions involve the rearrangement of electrons, leading to the formation of new compounds.Atoms of the same element can have different numbers of neutrons, resulting in isotopes.Electrons do not follow precise paths but exist in regions of high probability called orbitals.Protons and neutrons are located in the nucleus at the center of the atom, while electrons occupy specific energy levels or orbitals around the nucleus.Atoms are composed of subatomic particles, including protons, neutrons, and electrons.Modern Atomic Theory, also known as the Quantum Mechanical Model of the Atom, has built upon Dalton's theory and incorporates new discoveries and advancements in the field of atomic physics. However, as scientific knowledge advanced, it became evident that some of Dalton's postulates were oversimplified or inaccurate. Chemical reactions involve the rearrangement of atoms, but no atoms are created, destroyed, or changed into atoms of another element.ĭalton's theory provided a framework for understanding the behavior of matter and explaining various chemical phenomena.Atoms of different elements can combine in simple whole number ratios to form compounds.Atoms of the same element are identical in size, mass, and other properties.All matter is composed of indivisible particles called atoms.Dalton's Atomic Theoryĭalton's Atomic Theory, proposed in 1803, was a groundbreaking concept that revolutionized the field of chemistry. In this article, we will compare the attributes of Dalton's Atomic Theory with the advancements made in Modern Atomic Theory. Dalton's Atomic Theory laid the foundation for our understanding of atoms, but it has since been refined and expanded upon by modern atomic theory. 
One of the earliest atomic theories was proposed by John Dalton in the early 19th century. The study of atoms and their behavior has evolved significantly over the years.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
